 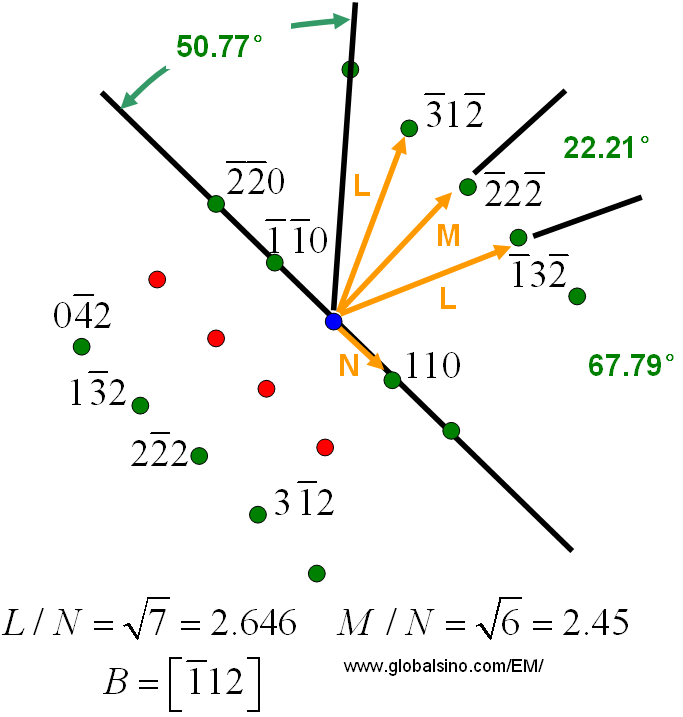
Make a plot of closest approach distance versus number fraction zinc, at 0 this is the distance for pure copper determined above and at 1.0 this is the number determined above for copper.Determine the distance of approach for the two patterns ( for HCP this is "a", for FCC this is "a"/ √ (2).Index both crystal structures and determine the lattice parameters.Measure the diffractometer trace for copper and zinc.In the powder mixture a calculation of the relative intensity of two peaks will be used to determine the amount of BCC Moly and FCC copper in a mixture containing independent crystals of these two materials. Vegard's Law will be used to determine the zinc content by consideration of the shift in the lattice parameter for the FCC pattern. This lab will involve the analysis of composition of two samples, a brass key, composed of a substitutional solution of zinc in copper (FCC) and a physical mixture of copper (FCC) and molybdenum (BCC) powder. 139 section 4-12 and equation 4-21 for Diffractometer. 12-6, Chapter 14 (Phase Analysis), Chapter 7 (Diffractometer), pp. New Cullity and Stock: Chapter 11, Figure 11-6, Chapter 12 (PhaseĪnalysis), Chapter 6 (Diffractometer), Chapter 4 (Calculated Diffracted Intensity) Objective: To become familiar with use of the diffractometer for quantitative analysis of phases in mixtures/alloys of metals. Here you will find options to view and activate subscriptions, manage institutional settings and access options, access usage statistics, and more.Lab 6: Diffractometer/Phase Analysis & CompositionĬopper Wire (oriented), Copper Plate (unoriented), Zinc Powder, Brass Key, Moly Powder, Moly/Copper Powder If you believe you should have access to that content, please contact your librarian.įor librarians and administrators, your personal account also provides access to institutional account management. The institutional subscription may not cover the content that you are trying to access. Oxford Academic is home to a wide variety of products. View the institutional accounts that are providing access.View your signed in personal account and access account management features.Some societies use Oxford Academic personal accounts to provide access to their members.Ĭlick the account icon in the top right to: See below.Ī personal account can be used to get email alerts, save searches, purchase content, and activate subscriptions. Some societies use Oxford Academic personal accounts to provide access to their members. If you do not have a society account or have forgotten your username or password, please contact your society. Do not use an Oxford Academic personal account. When on the society site, please use the credentials provided by that society.If you see ‘Sign in through society site’ in the sign in pane within a journal: Many societies offer single sign-on between the society website and Oxford Academic. 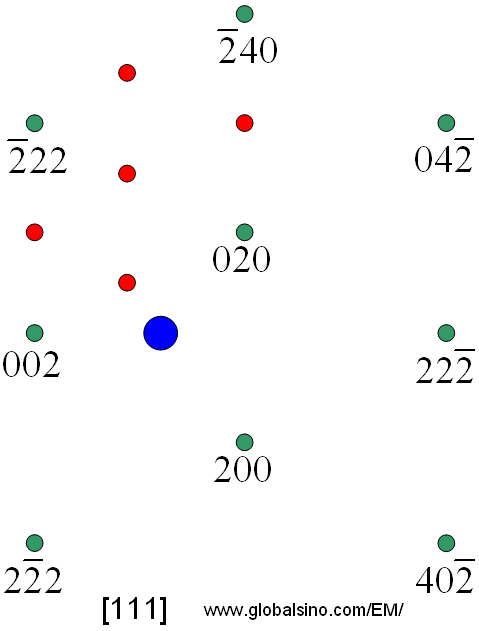
Society member access to a journal is achieved in one of the following ways: If you cannot sign in, please contact your librarian. If your institution is not listed or you cannot sign in to your institution’s website, please contact your librarian or administrator.Įnter your library card number to sign in. Following successful sign in, you will be returned to Oxford Academic.When on the institution site, please use the credentials provided by your institution.Select your institution from the list provided, which will take you to your institution's website to sign in.Click Sign in through your institution.Shibboleth / Open Athens technology is used to provide single sign-on between your institution’s website and Oxford Academic. This authentication occurs automatically, and it is not possible to sign out of an IP authenticated account.Ĭhoose this option to get remote access when outside your institution. Typically, access is provided across an institutional network to a range of IP addresses. If you are a member of an institution with an active account, you may be able to access content in one of the following ways: Get help with access Institutional accessĪccess to content on Oxford Academic is often provided through institutional subscriptions and purchases. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
